
Chemistry, 11.04.2021 20:10 michaelhal4271
(c) Write the balanced neutralization reaction and calculate the volume in mL of a 1.420 M NaOH solution required to titrate 25.00 mL of a 5.400 M H2SO4 solution

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, bibhu42kumarp7o4ss
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 20:30, allofthosefruit
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3


Chemistry, 23.06.2019 07:00, mahogany1956
What is the difference between covalent bonds and ionic bonds? covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the electrical attraction between charged atoms. covalent bonds involve the transfer of electrons between charged atoms; ionic bonds involve the sharing of electrons between atoms. covalent bonds involve the sharing of pairs of electrons between atoms; ionic bonds involve the sharing of single electrons between atoms. covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the sharing of protons between charged atoms.
Answers: 1
You know the right answer?
(c) Write the balanced neutralization reaction and calculate the volume in mL of a 1.420 M NaOH solu...
Questions in other subjects:

Mathematics, 05.05.2020 08:06

Biology, 05.05.2020 08:06



Mathematics, 05.05.2020 08:06

History, 05.05.2020 08:06

English, 05.05.2020 08:06



History, 05.05.2020 08:06

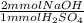 = 270 mmol NaOH
= 270 mmol NaOH

