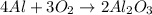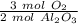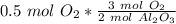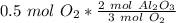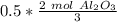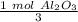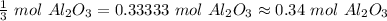Chemistry, 10.04.2021 06:40 sharondot2398
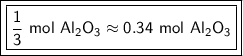
How many moles of aluminum oxide will be produced from 0.50 mol of oxygen? 4 Al + 3 O2→ 2 Al2O3

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 18:00, darg3990rgp2t0r2
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3

Chemistry, 21.06.2019 23:00, jamccoy3335
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 01:30, lizethdominguez037
What is the value of keq for the reaction expressed in scientific notation
Answers: 1
You know the right answer?
How many moles of aluminum oxide will be produced from 0.50 mol of oxygen?
4 Al + 3 O2→ 2 Al2O3...
Questions in other subjects: