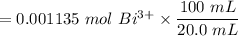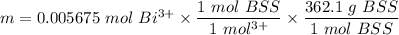Chemistry, 08.04.2021 22:20 adayisenga
The tablets were crushed, and 4.9993 g of the powder was transferred to a beaker and reacted with HCl. After filtration, the filtrate was transferred to a 100-mL volumetric flask and diluted with water. 20.00 mL of this stock solution were combined with 0.2 M Na3PO4. The resulting precipitate weighed 0.3451 g after drying. Calculate the moles of BiPO4 precipitated, the moles of Bi3 in the stock solution, and the mass of BSS per tablet.

Answers: 1


Other questions on the subject: Chemistry




Chemistry, 22.06.2019 21:30, thompsonhomes1
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3
You know the right answer?
The tablets were crushed, and 4.9993 g of the powder was transferred to a beaker and reacted with HC...
Questions in other subjects:

English, 08.09.2019 21:20

Mathematics, 08.09.2019 21:20

Mathematics, 08.09.2019 21:20




Mathematics, 08.09.2019 21:20

Mathematics, 08.09.2019 21:20

Physics, 08.09.2019 21:20