
Chemistry, 05.04.2021 19:10 angienunez2983
See all photos
Share
A
+ Add to
% Edit & Create
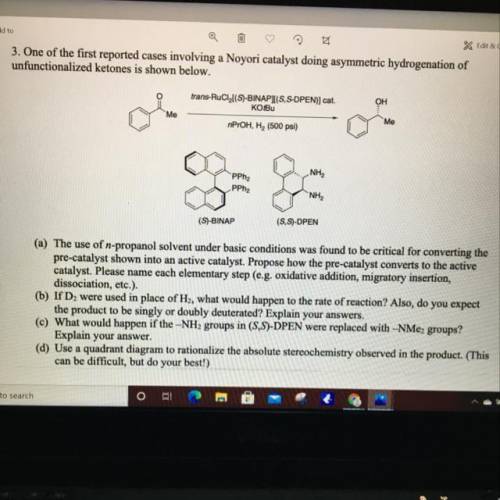
3. One of the first reported cases involving a Noyori catalyst doing asymmetric hydrogenation of
unfunctionalized ketones is shown below.
trans-RuCl(S)-BINAPI(S, S-DPEN)] cat.
KOBU
OH
Me
Me
nPrOH, H2 (500 psi)
NH2
PPha
PPha
"NH2
(S)-BINAP
(SS)-DPEN
(a) The use of n-propanol solvent under basic conditions was found to be critical for converting the
pre-catalyst shown into an active catalyst. Propose how the pre-catalyst converts to the active
catalyst. Please name each elementary step (e. g. oxidative addition, migratory insertion,
dissociation, etc.).
(b) If D2 were used in place of H2, what would happen to the rate of reaction? Also, do you expect
the product to be singly or doubly deuterated? Explain your answers.
(c) What would happen if the -NH2 groups in (S, S)-DPEN were replaced with -NMe2 groups?
Explain your answer.
(d) Use a quadrant diagram to rationalize the absolute stereochemistry observed in the product. (This
can be difficult, but do your best!)

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:10, cheesedoodle
When a nucleus absorbs a neutron and then breaks apart, there are many products of the reaction. what is not a product of a nuclear fission reaction
Answers: 1

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3


Chemistry, 22.06.2019 18:30, lattimorekeonna1
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1
You know the right answer?
See all photos
Share
A
+ Add to
% Edit & Create
3. One of the first rep...
A
+ Add to
% Edit & Create
3. One of the first rep...
Questions in other subjects:


Mathematics, 06.05.2020 04:09



Business, 06.05.2020 04:09

History, 06.05.2020 04:09



Mathematics, 06.05.2020 04:09



