Chemistry, 01.04.2021 21:40 kaywendel2008
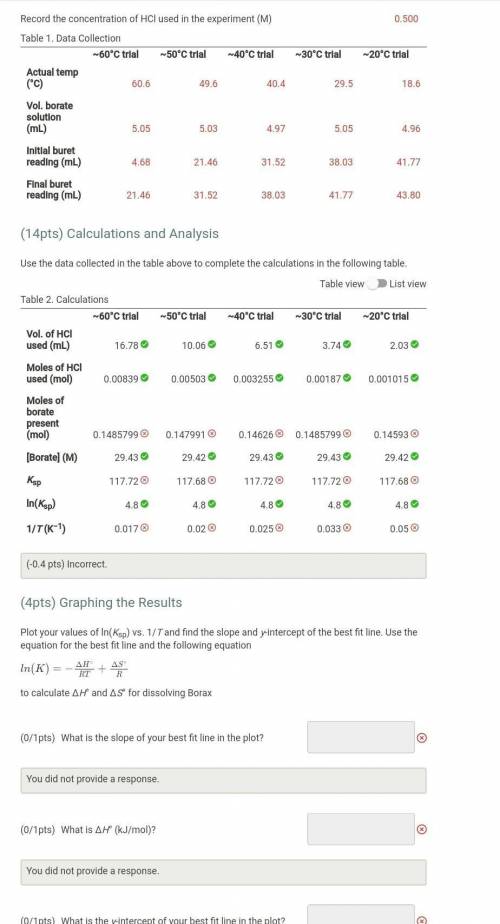
Plot your values of ln(Ksp) vs. 1/T and find the slope and y-intercept of the best fit line. Use the equation for the best fit line and the following equation
to calculate ΔH° and ΔS° for dissolving Borax
(0/1pts)
What is the slope of your best fit line in the plot?
at is ΔH° (kJ/mol)?
highlight_off
(0/1pts)
What is the y-intercept of your best fit line in the plot?
highlight_off
(-1 pts)
(0/1pts)
What is ΔS° (J/mol)?
l

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:50, amandamac7339
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 23.06.2019 03:30, jennelledenise
Mr. rose asked his student to draw a quadrilateral with four unequal sides. an example of this kind of quadrilateral
Answers: 1

You know the right answer?
Plot your values of ln(Ksp) vs. 1/T and find the slope and y-intercept of the best fit line. Use the...
Questions in other subjects:

Mathematics, 22.04.2021 01:00

Chemistry, 22.04.2021 01:00



Mathematics, 22.04.2021 01:00