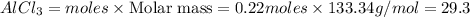PLEASE HELP
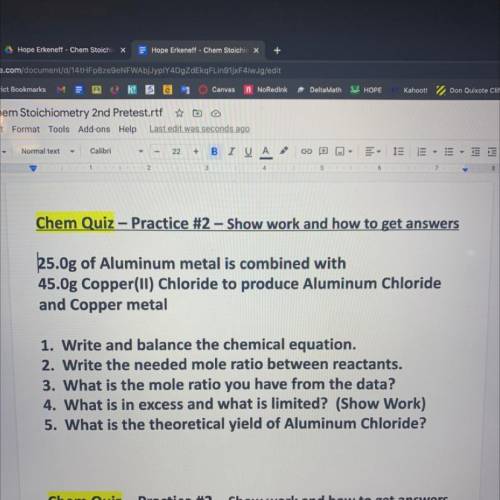
25.0g of Aluminum metal is combined with
45.Og Copper(ll) Chloride to produce Alu...

PLEASE HELP
25.0g of Aluminum metal is combined with
45.Og Copper(ll) Chloride to produce Aluminum Chloride
and Copper metal
1. Write and balance the chemical equation.
2. Write the needed mole ratio between reactants.
3. What is the mole ratio you have from the data?
4. What is in excess and what is limited? (Show Work)
5. What is the theoretical yield of Aluminum Chloride?

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:10, coastieltp58aeg
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 22:00, huddyxo
Scientists often have to deal with numbers that are either very large or very small. for example, the radius of the sun is approximately 696,000 kilometers, while bacterial cells are as small as 1.9 × 10-4 millimeters. express each number in an alternate form.
Answers: 1

Chemistry, 22.06.2019 22:30, xlebrny7831
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
Questions in other subjects:

Mathematics, 24.06.2021 14:00

Mathematics, 24.06.2021 14:00

Biology, 24.06.2021 14:00

Mathematics, 24.06.2021 14:00

History, 24.06.2021 14:00


Chemistry, 24.06.2021 14:00



English, 24.06.2021 14:00

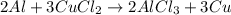
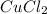 : 2 moles of
: 2 moles of 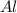
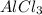 is 29.3 g
is 29.3 g 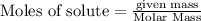
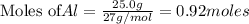
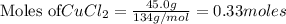
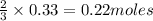 of
of