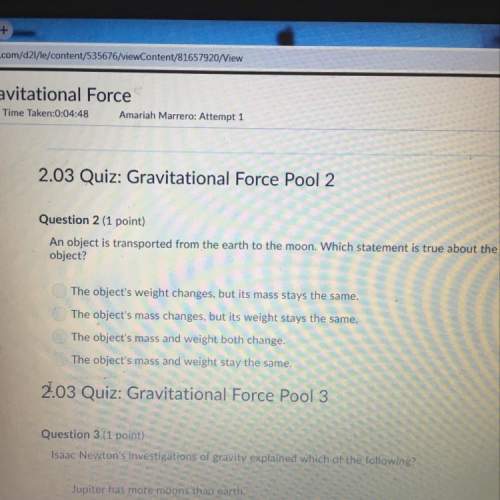
Chemistry, 31.03.2021 22:50 genyjoannerubiera
For the production of ammonia reaction seen below, Hydrogen is
the limiting reagent. First, determine how much ammonia you can
produce if you start with 18.4 g of hydrogen. Then determine your
percent yield for this reaction if the actual yield produced in the lab
was 84.7 grams.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:00, bettybales1986
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 22.06.2019 09:30, janetexcoelho
What does the mass of 0.7891 mol of ferric oxide (fe2o3)
Answers: 1


Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1
You know the right answer?
For the production of ammonia reaction seen below, Hydrogen is
the limiting reagent. First, determi...
Questions in other subjects:




Mathematics, 06.03.2020 01:02


Mathematics, 06.03.2020 01:02

Mathematics, 06.03.2020 01:02