WILL AWARD BRAINLIEST!!
...

Chemistry, 31.03.2021 04:30 laylay2380
WILL AWARD BRAINLIEST!!
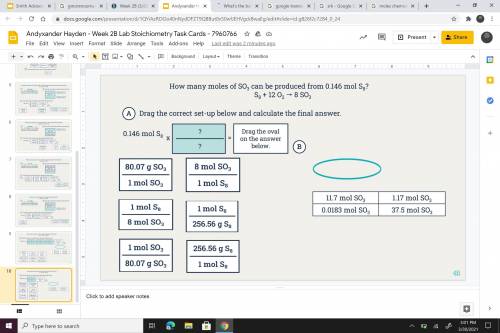

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 20.06.2019 18:04, augestfaith
Which stage of the cell cycle usually takes the longest?
Answers: 3

Chemistry, 22.06.2019 03:30, ruleolivas
Asample of ammonia reacts with oxygen as shown. 4nh3(g) + 5o2(g) 4no(g) + 6h2o(g) what is the limiting reactant if 4.0 g of nh3 react with 8.0 g of oxygen? o2 because it produces only 0.20 mol of no. nh3 because it produces only 0.20 mol of no. o2 because it produces two times less no than nh3. nh3 because it produces three times more no than o2.
Answers: 3

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

You know the right answer?
Questions in other subjects:

Mathematics, 20.01.2020 01:31


English, 20.01.2020 01:31


Mathematics, 20.01.2020 01:31

Mathematics, 20.01.2020 01:31






