

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:30, hdhtvthjr
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 14:00, leahstubbs
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3

Chemistry, 22.06.2019 19:00, montgomerykarloxc24x
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 22.06.2019 19:30, liyahlanderson2232
Astring vibrates with a frequency of 10 hz. why can't a person hear the sound waves produced by the vibrating string, no matter how large the amplitude of the waves? out! this is homework and due tomorrow! you so much!
Answers: 2
You know the right answer?
A sealed 1.0L flask is filled with 0.500 mols of I_2 and 0.500 mols of Br_2. When the container achi...
Questions in other subjects:

Social Studies, 05.10.2021 17:00


Mathematics, 05.10.2021 17:00

Physics, 05.10.2021 17:00



Mathematics, 05.10.2021 17:00

Mathematics, 05.10.2021 17:00

Mathematics, 05.10.2021 17:00

Arts, 05.10.2021 17:00

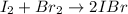
![K=\frac{[IBr]^2}{[I_2][Br_2]} =0.0110](/tpl/images/1228/6485/945be.png)
 (reaction extent) would be:
(reaction extent) would be: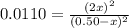
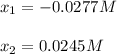
![[IBr]=2x=2*0.0249M=0.049M](/tpl/images/1228/6485/0c444.png)


