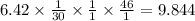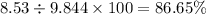Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 6.42 grams of nitrogen monoxide are mixed with excess oxygen gas . The reaction yields 8.53 grams of nitrogen dioxide . nitrogen monoxide ( g ) oxygen ( g ) nitrogen dioxide ( g ) What is the theoretical yield of nitrogen dioxide

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:00, luffybunny
There is one girl i like and i don't know how to tell her that, i have a feeling she knows but if she doesn't i don't want to make a fool out of myself how is one way to boost my confidence on asking her out
Answers: 1


Chemistry, 22.06.2019 21:00, agarcia24101993
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
Nitrogen monoxide is produced by combustion in an automobile engine. For the following reaction, 6.4...
Questions in other subjects:

Mathematics, 07.07.2019 02:10



English, 07.07.2019 02:10