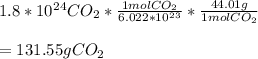Chemistry, 29.03.2021 07:00 khanhlan7213
How many grams of CO2 (Molar Mass = 44.01 grams) are in 1.8x1024 molecules of CO2 ?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2


You know the right answer?
How many grams of CO2 (Molar Mass = 44.01 grams) are in 1.8x1024 molecules of CO2 ?...
Questions in other subjects:

Biology, 28.07.2019 14:30


Chemistry, 28.07.2019 14:30

Chemistry, 28.07.2019 14:30

History, 28.07.2019 14:30

Social Studies, 28.07.2019 14:30

Biology, 28.07.2019 14:30

History, 28.07.2019 14:30