
Rank the following carboxylic acids by acid strength, with the strongest at the top and the weakest at the bottom. It may help to draw each Lewis structure. Drag and drop options into correct order. For keyboard navigation...SHOW MORE Press space or enter to grab A. CF3CO2H B. CHF2CO2HC. CH2FCO2H D. CH3CO2H

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, clairebear66
What three natural resources are found in the great lakes region
Answers: 2

Chemistry, 22.06.2019 08:00, wizz4865
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 09:00, kcarstensen59070
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1
You know the right answer?
Rank the following carboxylic acids by acid strength, with the strongest at the top and the weakest...
Questions in other subjects:



English, 28.06.2019 07:30


Biology, 28.06.2019 07:30

History, 28.06.2019 07:30

History, 28.06.2019 07:30

Mathematics, 28.06.2019 07:30


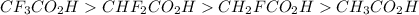
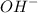 groups in the COOH group towards itself, making it relatively easy to loose the proton of the carboxyl group.
groups in the COOH group towards itself, making it relatively easy to loose the proton of the carboxyl group.

