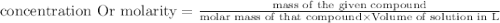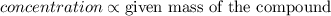James is preparing three solutions of salt water. each of the solutions has the same amount of water in the beaker and a different amounts of salt. in the first beaker, there is one scoop of salt in the beaker. the second beaker has two scoops of salt. the third beaker has three scoops of salt. which of the beakers is the most concentrated solution?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:50, Jerrikasmith28
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2

Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 07:40, caleb19moody
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2
You know the right answer?
James is preparing three solutions of salt water. each of the solutions has the same amount of water...
Questions in other subjects:

Mathematics, 28.01.2021 18:30


Mathematics, 28.01.2021 18:30

Physics, 28.01.2021 18:30

Mathematics, 28.01.2021 18:30

Mathematics, 28.01.2021 18:30


Mathematics, 28.01.2021 18:30

Arts, 28.01.2021 18:30