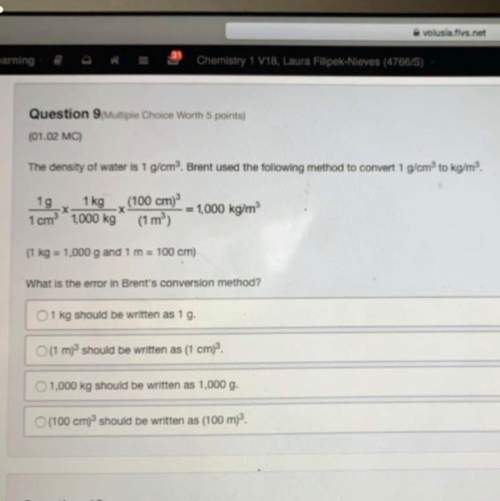
We have seen that adding the common ion (salt of the conjugate base) to a weak acid equilibrium changes the pH. Imagine a solution in which you have created a solution that is 2.0 M in acetic acid and 2.0 M in sodium acetate (the purple atom is the sodium cation). In this case the concentration of the weak acid and it's conjugate base are exactly equal. In truth, a small amount of acetate anion (conjugate base) is also coming from the dissociation of the weak acid, but this is negligible compared to the added sodium acetate.
a. The Na+ cation does not appear in the equilibrium because it is a spectator ion and is only the counter ion for the acetate anion.
b. All of the acetic acid will become deprotonated by water so the final solution contains ony aqueous h aron un ions and acetate ons.
c. The pKa for the acid = the pH of this solution. Since acetic acid is a weak acid, acetate anion is considered to be a strong base.
d. The [H3O+]=Ka in this solution. The pH of this solution will be lower than that of a 2.0 M solution of acetic acid.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, hdjsjfjruejchhehd
Define dew point. i am writing this part to be able to ask the question
Answers: 1

Chemistry, 22.06.2019 12:40, carebear60
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1
You know the right answer?
We have seen that adding the common ion (salt of the conjugate base) to a weak acid equilibrium chan...
Questions in other subjects:

Mathematics, 22.05.2021 04:20



Mathematics, 22.05.2021 04:20

Business, 22.05.2021 04:20

Chemistry, 22.05.2021 04:20



Arts, 22.05.2021 04:20

Mathematics, 22.05.2021 04:20