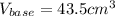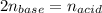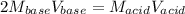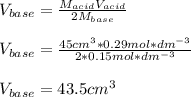Chemistry, 22.03.2021 02:10 juansantos7b
Calculate the volume of a 0.15 mol dm-3 Ba(OH)2 solution required to completely neutralize 45 cm3 of a 0.29 mol dm-3 HNO3 solution. Note: Ba(OH)2 + 2HNO3 --> Ba(NO3)2 + 2H2O

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:00, kkmonsterhigh18
The diagram below shows a cell placed in a solution. a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution. only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it. it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 09:30, junkmailemail42
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

You know the right answer?
Calculate the volume of a 0.15 mol dm-3 Ba(OH)2 solution required to completely neutralize 45 cm3 of...
Questions in other subjects:

History, 12.04.2021 18:10

Mathematics, 12.04.2021 18:10


Physics, 12.04.2021 18:10

Mathematics, 12.04.2021 18:10


History, 12.04.2021 18:10

Mathematics, 12.04.2021 18:10