Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 12:30, nekathadon
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1

You know the right answer?
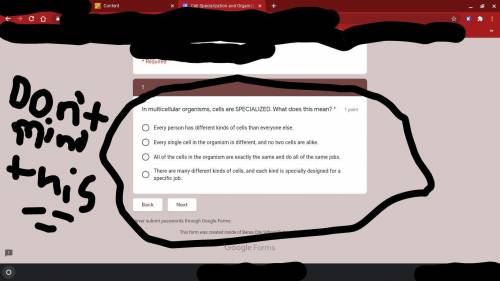
I might quit because people are just using me for my points. could someone answer this for me?
Questions in other subjects:



Mathematics, 20.11.2020 01:00

Mathematics, 20.11.2020 01:00

History, 20.11.2020 01:00

Mathematics, 20.11.2020 01:00



Mathematics, 20.11.2020 01:00