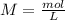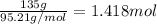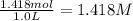Chemistry, 18.03.2021 01:30 markrandsdenn
If 135 grams of MgCl2 are dissolved in a 1.0 L solutions, what is the molarity of the solution?

Answers: 2


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 20:00, jalenevoyles
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3

Chemistry, 23.06.2019 01:00, ZaNiyahlove4711
Which of the following is the molecular formula for a simple sugar? a. cooh b. h2o c. oh d. c6h12o6
Answers: 1
You know the right answer?
If 135 grams of MgCl2 are dissolved in a 1.0 L solutions, what is the molarity of the solution?...
Questions in other subjects:





Mathematics, 30.07.2020 01:01