
Chemistry, 18.03.2021 01:20 alondra6190
Nitrogen dioxide gas is dark brown in color and remains in equilibrium with dinitrogen tetroxide gas, which is colorless.
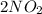 (g) ⇌
(g) ⇌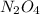 (g)
(g)
When the mixture was moved from room temperature to a higher temperature, the mixture turned darker brown. Which of the following conclusions about the mixture is true?
A. The forward reaction is exothermic.
B. The backward reaction has a negative net enthalpy.
C. The colorless gas has a higher enthalpy than the brown gas.
I know that is an exothermic reaction, but it shifts to left, so it should be a backward reaction, not a forward. Therefore, I don't think it is A. I am leaning towards B, but I would like someone to double-check my thinking. Please no fake answers or external links. Thanks in advance!

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, ayaanwaseem
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

You know the right answer?
Nitrogen dioxide gas is dark brown in color and remains in equilibrium with dinitrogen tetroxide gas...
Questions in other subjects:



Biology, 21.09.2019 06:30

Social Studies, 21.09.2019 06:30

English, 21.09.2019 06:30

History, 21.09.2019 06:30

Mathematics, 21.09.2019 06:30

Mathematics, 21.09.2019 06:30




