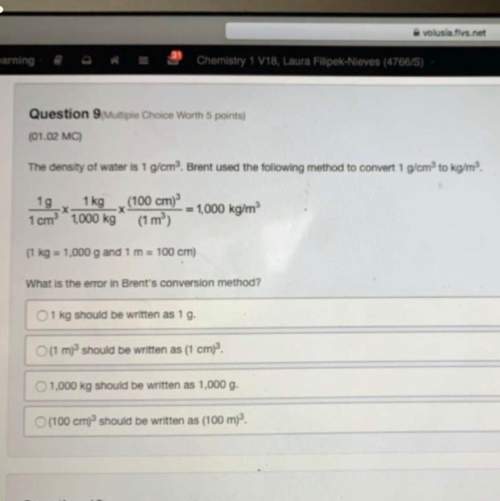
Select the correct answer.
An iron bar at 200*C is placed in thermal contact with an identical iron bar at 120°C in an isolated system. After 30 minutes, both iron bars are at 160°C. If the iron bars were placed
in thermal contact in an open system instead of an isolated system, how would
he results be different? Assume that the room temperature is 25°C.
The temperatures of the iron bars after 30 minutes would be less than 160°C because heat would be lost to the surroundings.
B. It would take more than 30 minutes for both iron bars to reach 160°C
to reach 160°C because heat would be transferred less efficiently.
C.
The temperatures of both iron bars would increase as they absorb heat from the surroundings.
D.
The temperatures of both iron bars would decrease because pieces of them would be lost to the surroundings.

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:30, charles8527
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2

Chemistry, 22.06.2019 15:20, shanyeah
Water is initially present in a state where its molecules are far apart. during a change of state, its molecules slow down. which change of state has most likely taken place? from a gas to a liquid from a liquid to a gas from a solid to a liquid from a gas to a plasma
Answers: 1

Chemistry, 23.06.2019 01:30, nikonee
Astudent states that 9.0 g of baking soda will form an unsaturated solution in 100 g of water. what do you need to know to decide whether this statement is correct? a. the temperature of the water and the molar mass of baking soda b. the percent by volume of the solution and the solubility of baking soda c. the temperature of the water and the solubility of baking soda at that temperature
Answers: 1

You know the right answer?
Select the correct answer.
An iron bar at 200*C is placed in thermal contact with an identical iron...
Questions in other subjects:


English, 21.07.2019 02:30

Mathematics, 21.07.2019 02:30



Mathematics, 21.07.2019 02:30


Chemistry, 21.07.2019 02:30