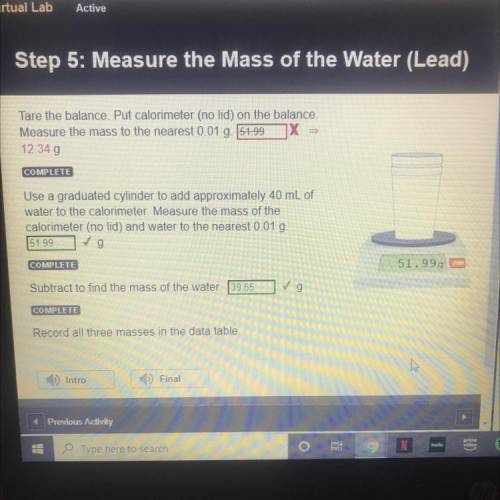
STEP 5: LEAD
Tare the balance. Put calorimeter (no lid) on the balance.
Measure the mass to t...

Chemistry, 17.03.2021 23:50 bartekpiglo
STEP 5: LEAD
Tare the balance. Put calorimeter (no lid) on the balance.
Measure the mass to the nearest 0.01 g.
12.34 g
Use a graduated cylinder to add approximately 40 mL of
water to the calorimeter. Measure the mass of the
calorimeter (no lid) and water to the nearest 0.01 g.
51.99 g
Subtract to find the mass of the water. 39.65 g

Answers: 3


Other questions on the subject: Chemistry



Chemistry, 22.06.2019 04:00, queenkimm26
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3
You know the right answer?
Questions in other subjects:

Mathematics, 13.03.2020 01:01

Mathematics, 13.03.2020 01:01


History, 13.03.2020 01:01


Social Studies, 13.03.2020 01:01

Mathematics, 13.03.2020 01:01






