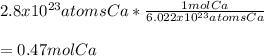b. 0.47 moles Ca

Chemistry, 11.03.2021 08:10 sassyparker1216
How many moles are in 2.8x10^23 atoms of Calcium?
a. 6.98 moles Ca
b. 0.47 moles Ca
c. 1.1x10^25 moles Ca
d. 1.7x10^47 moles Ca

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Aidanjsauer
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2


Chemistry, 22.06.2019 19:00, ghernadez
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1

Chemistry, 23.06.2019 01:30, Dmoney5104
The biomedical technique in which a part of the brain is destroyed with electric current is known as a. electroconvulsive therapy b. prefrontal lobotomy c. bilateral cingulotomy d. tardive dyskinesia
Answers: 2
You know the right answer?
How many moles are in 2.8x10^23 atoms of Calcium?
a. 6.98 moles Ca
b. 0.47 moles Ca
b. 0.47 moles Ca
Questions in other subjects:



History, 24.07.2019 04:00

Mathematics, 24.07.2019 04:00


Health, 24.07.2019 04:00

Mathematics, 24.07.2019 04:00