
Chemistry, 09.03.2021 21:10 mxrvin4977
2020-21 CCS 8SCI SMA 10 P.1.1, 1.22 of 18 Atoms of elements X and Y react to form a compound. The atoms present before the reaction and the atoms left unreacted after the reaction are shown below.
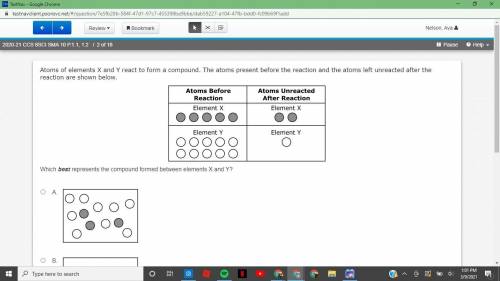
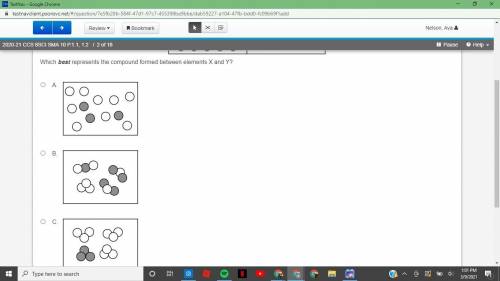
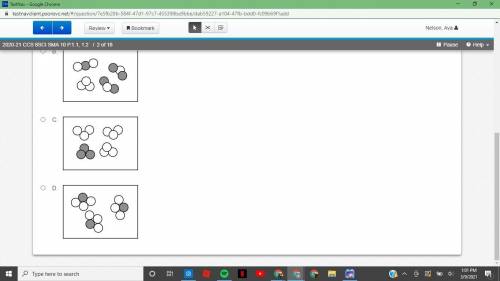

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, Apple557
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1


Chemistry, 22.06.2019 12:40, whitethunder05
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 16:00, bbrogle5154
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1
You know the right answer?
2020-21 CCS 8SCI SMA 10 P.1.1, 1.22 of 18
Atoms of elements X and Y react to form a compound. The a...
Questions in other subjects:


Chemistry, 05.10.2020 09:01

Chemistry, 05.10.2020 09:01

World Languages, 05.10.2020 09:01


English, 05.10.2020 09:01

Mathematics, 05.10.2020 09:01

Physics, 05.10.2020 09:01

Mathematics, 05.10.2020 09:01



