
Chemistry, 09.03.2021 05:50 NutMeg6544
The equilibrium constant, Kc, is calculated using molar concentrations. For gaseous reactions another form of the equilibrium constant, Kp, is calculated from partial pressures instead of concentrations. These two equilibrium constants are related by the equation Kp=Kc(RT)Δn where R=0.08206 L⋅atm/(K⋅mol), T is the absolute temperature, and Δn is the change in the number of moles of gas (sum moles products - sum moles reactants). For example, consider the reaction N2(g)+3H2(g)⇌2NH3(g) for which Δn=2−(1+3)=−2. Part A For the reaction 2A(g)+3B(g)⇌C(g) Kc = 63.2 at a temperature of 81 ∘C . Calculate the value of Kp. Express your answer numerically.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:30, raizagisselle1694
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

You know the right answer?
The equilibrium constant, Kc, is calculated using molar concentrations. For gaseous reactions anothe...
Questions in other subjects:


Mathematics, 26.12.2019 03:31

Computers and Technology, 26.12.2019 03:31


Computers and Technology, 26.12.2019 04:31





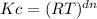 = (0.082 L.atm/K.mol x 354 K)⁻⁴ = 1.41 x 10⁻⁶
= (0.082 L.atm/K.mol x 354 K)⁻⁴ = 1.41 x 10⁻⁶


