Chemistry, 09.03.2021 04:30 lexibyrd120
gA scientist is trying to discover information about an unknown metal in a compound. The formula for the compound is believed to be XBr3XBr3 where XX is the unknown metal. The scientist determined that a 4.700 g4.700 g sample of this compound contains 4.834×10−2 mol Br4.834×10−2 mol Br . Calculate the atomic mass of the unknown metal, XX .

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:50, ajaydonlee
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 09:00, tashaunalewis4786
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1

Chemistry, 22.06.2019 17:40, Snowball080717
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3
You know the right answer?
gA scientist is trying to discover information about an unknown metal in a compound. The formula for...
Questions in other subjects:

Social Studies, 05.04.2020 21:29


Mathematics, 05.04.2020 21:30



Mathematics, 05.04.2020 21:30

Mathematics, 05.04.2020 21:30


Mathematics, 05.04.2020 21:30

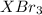
 mol of Br.
mol of Br.  contain in 1 mol of
contain in 1 mol of 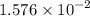 mol of
mol of 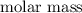 of
of