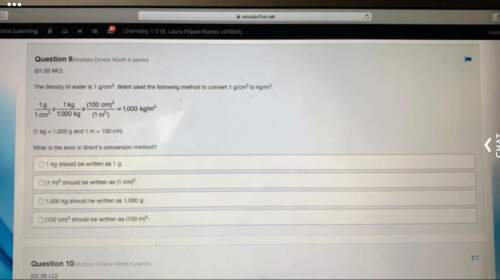
Chemistry, 07.03.2021 02:20 stricklandjoseph12
One of the primary substances used in sandpaper is Silicon carbide, SiC. It is produced by heating silicon dioxide, SiO2, with Carbon in a high intensity furnace. The other product is carbon monoxide, CO. What is the percent yield when 202 grams of SiC is produced from the reaction of 334.5 grams of carbon with 427 grams of SiO2? SiO2 (s) + C (s) → SiC (s) + CO (g) (unbalanced equation)

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, isabelvaldez123
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 20:30, lexibyrd120
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 22.06.2019 23:40, sydneykated
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
You know the right answer?
One of the primary substances used in sandpaper is Silicon carbide, SiC. It is produced by heating s...
Questions in other subjects:

Mathematics, 12.01.2021 23:30