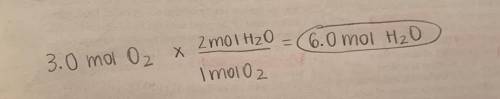Use the balanced equation below
O2 + 2H2 > 2H2O
1. How many moles of hydrogen are ne...

Chemistry, 07.03.2021 01:00 markarianlaura1
Use the balanced equation below
O2 + 2H2 > 2H2O
1. How many moles of hydrogen are needed to complete the reaction?
2. If 3.0 moles of oxygen are present with excess hydrogen, how many moles of water are formed?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, pressure772
Which is a character of nuclear fusion but not nuclear fission
Answers: 3

Chemistry, 22.06.2019 07:20, camillexv2668
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 22.06.2019 10:00, aschool2000
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
You know the right answer?
Questions in other subjects:







Social Studies, 05.01.2021 16:50


Mathematics, 05.01.2021 16:50