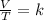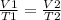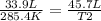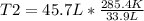Chemistry, 06.03.2021 05:20 cmflores3245
At what temperature would the volume of a gas be equal to 45.7 L, if the volume of that gas was 33.9 L at 12.4ºC? 241.9 K 241.9 K 451.2 K 451.2 K 384.7 K 384.7 K 273.15 K

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:20, aidengalvin20
One or more substances changing into one or more substances is an example of a
Answers: 1

Chemistry, 22.06.2019 21:50, donttrip10
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state. a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2

Chemistry, 23.06.2019 01:50, UncleVictor5188
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1
You know the right answer?
At what temperature would the volume of a gas be equal to 45.7 L, if the volume of that gas was 33.9...
Questions in other subjects:



History, 17.07.2019 21:30

History, 17.07.2019 21:30


History, 17.07.2019 21:30

Social Studies, 17.07.2019 21:30

History, 17.07.2019 21:30