
Chemistry, 04.03.2021 21:00 AkramMasoud
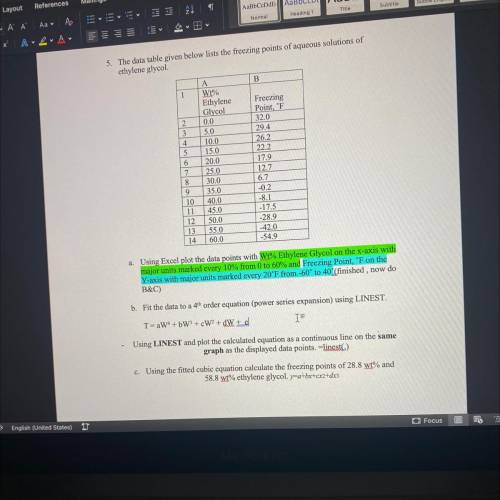
5. The data table given below lists the freezing points of aqueous solutions of
ethylene glycol.
B
1
2
3
4
5
6
7
8
9
10
11
12
13
14
A
Wi%
Ethylene
Glycol
0.0
5.0
10.0
15.0
20.0
25.0
30.0
35.0
40.0
45.0
50.0
55.0
60.0
Freezing
Point, 'F
32.0
29.4
26.2
22.2
17.9
12.7
6.7
-0.2
-8.1
-17.5
-28.9
-42.0
-54.9
a. Using Excel plot the data points with Wi% Ethylene Glycol on the x-axis with
major units marked every 10% from 0 to 60% and Freezing Point, 'F on the
Y-axis with major units marked every 20°F from -60° to 40(finished, now do
B&C)
b. Fit the data to a 4h order equation (power series expansion) using LINEST.
T-aW4 +6W + cW2 + dW+d
IF
Using LINEST and plot the calculated equation as a continuous line on the same
graph as the displayed data points. -linest)
c. Using the fitted cubic equation calculate the freezing points of 28.8 wt% and
58.8 wt% ethylene glycol. yua+bx+cx2+x3
K

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, ReveenatheRaven2296
Which type of reaction always has an element and a compound as reactants
Answers: 1

Chemistry, 22.06.2019 04:00, eborkins
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Chemistry, 22.06.2019 09:00, dante766
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1
You know the right answer?
5. The data table given below lists the freezing points of aqueous solutions of
ethylene glycol.
Questions in other subjects:



Social Studies, 12.12.2019 04:31

History, 12.12.2019 04:31


Mathematics, 12.12.2019 04:31


Mathematics, 12.12.2019 04:31

History, 12.12.2019 04:31



