
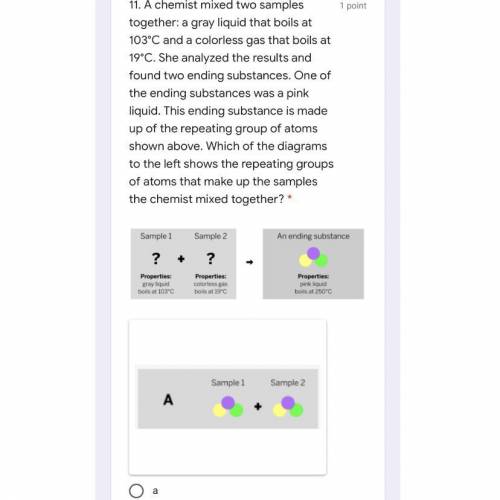
A chemist mixed two samples together: a gray liquid that boils at 103°C and a colorless gas that boils at 19°C. She analyzed the results and found two ending substances. One of the ending substances was a pink liquid. This ending substance is made up of the repeating group of atoms shown above. Which of the diagrams to the left shows the repeating groups of atoms that make up the samples the chemist mixed together?

Answers: 2


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, njones58emailtjcedu
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 18:30, tanviknawale
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1

Chemistry, 22.06.2019 19:00, ghernadez
Imagine that a new planet is discovered with two moons of equal mass: moon a and moon b. the mass of the new planet is greater than the combined mass of its moons. moon a is farther away from the new planet than moon b. what is the planet's gravitational pull on moon a compared to the planet's gravitational pull on moon b? the planet's gravity repels moon a with a greater force than it repels moon b, which is why moon a is farther away. the gravitational pull on moon b is greater than on moon a because moon b is closer to the new planet than moon a. the gravitational pull on moon b is greater than on moon a because moon b is farther away from the new planet than moon a. the gravitational pull on moon a is the same as the gravitational pull on moon b because distance does not affect the planet's gravity.
Answers: 1
You know the right answer?
A chemist mixed two samples together: a gray liquid that boils at 103°C and a colorless gas that boi...
Questions in other subjects:







Mathematics, 23.08.2021 21:10

Mathematics, 23.08.2021 21:10




