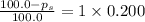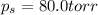Chemistry, 01.03.2021 22:00 lisilyn9755
What is the vapor pressure of a solution in which the mole fraction of the solute is 0.200 and the vapor pressure of the pure solvent is 100.0 torr? (Assume a single nonvolatile, nonelectrolyte solute).
a. 0 torr
b. 80.0 torr
c. 100.0 torr
d. 120.0 torr
e. 20.0 torr

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:00, tgraveslaylay2743
Bose-einstein condensation occurs at what temperature?
Answers: 2


Chemistry, 22.06.2019 01:40, georgehall3027
C3h8o3 - glycerol major species present when dissolved in water
Answers: 2
You know the right answer?
What is the vapor pressure of a solution in which the mole fraction of the solute is 0.200 and the v...
Questions in other subjects:



History, 03.03.2020 17:32



Mathematics, 03.03.2020 17:32

History, 03.03.2020 17:32



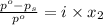
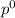 = vapor pressure of pure solvent = 100.0 torr
= vapor pressure of pure solvent = 100.0 torr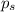 = vapor pressure of solution = ?
= vapor pressure of solution = ?
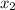 = mole fraction of solute = 0.200
= mole fraction of solute = 0.200