PLEASE HELP ASAP I WILL GIVE BRAINLIEST!
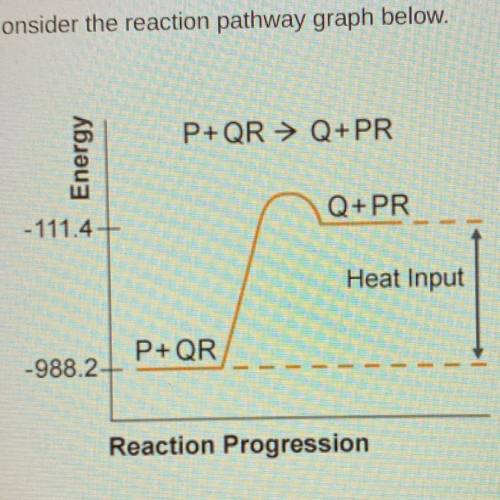
Consider the reaction pathway graph below.
The rate...

Chemistry, 01.03.2021 19:00 fixianstewart
PLEASE HELP ASAP I WILL GIVE BRAINLIEST!
Consider the reaction pathway graph below.
The rate increases by a factor of 9 when the concentration of A triples. The rate triples when the concentration of B triples. What is the new rate law for the reaction?
A) endothermic because Hrxn=-876.8 kJ
B) endothermic because Hrxn=876.8kJ
C)exothermic because Hrxn= -1099.6kJ
D) exothermic because Hrxn=1099.6kJ

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, skaterwolf1317
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 23:50, datboyjulio21
Which scientists contributed to the determination of how cfcs in clouds in the upper atmosphere could destroy ozone molecules
Answers: 1

You know the right answer?
Questions in other subjects:





History, 12.04.2021 18:10




Mathematics, 12.04.2021 18:10

Mathematics, 12.04.2021 18:10



