How many grams of CaO can be produced using 3.9 moles CaCO3?1
CaCO3 --> 1CaO + 1CO *...

Chemistry, 01.03.2021 14:00 SamaHafiz8984
How many grams of CaO can be produced using 3.9 moles CaCO3?1
CaCO3 --> 1CaO + 1CO *

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 23.06.2019 00:00, PineappleDevil889
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1

Chemistry, 23.06.2019 03:40, ElegantEmerald
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2

Chemistry, 23.06.2019 05:40, Queenquestion9130
Why is any chemical reaction always balanced? give reasons and explain the easiest way to solve the balancing problems in chemical equations with stoichiometric coefficients upto 20 as hit and trial doesn't always work. give full reasoning
Answers: 1
You know the right answer?
Questions in other subjects:



Biology, 15.07.2019 17:50

Biology, 15.07.2019 17:50




Geography, 15.07.2019 17:50


 will be produced from 3.9 moles of
will be produced from 3.9 moles of 

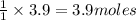 of
of 


