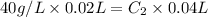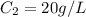Chemistry, 27.02.2021 15:30 samlolomg123
A solution has a concentration of 40 g. L-1
. We take 20 mL of this solution and add water. The
final volume is 40 mL. What's the new concentration? No calculations but you need to
justify

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, dustinquiz255
1) this is the structure in the cell nucleus that houses a cell's genetic information
Answers: 3


Chemistry, 22.06.2019 06:30, noathequeen
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

You know the right answer?
A solution has a concentration of 40 g. L-1
. We take 20 mL of this solution and add water. The
Questions in other subjects:

Mathematics, 17.06.2020 21:57



English, 17.06.2020 21:57



Mathematics, 17.06.2020 21:57



Mathematics, 17.06.2020 21:57

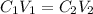
 = concentration of stock solution = 40 g/L
= concentration of stock solution = 40 g/L
 = volume of stock solution = 20 ml = 0.02 L
= volume of stock solution = 20 ml = 0.02 L
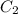 = concentration of diluted solution= ?
= concentration of diluted solution= ?
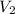 = volume of diluted solution= 40 ml = 0.04 L
= volume of diluted solution= 40 ml = 0.04 L