
Chemistry, 26.02.2021 22:50 Maxwell8066
Use CER format and label each part. Support your answers to question 15 and 16 with evidence. As part of your evidence include the number and types of the specific molecules before and after cooling as well as evidence of a chemical change. (Pls help xnnsx)
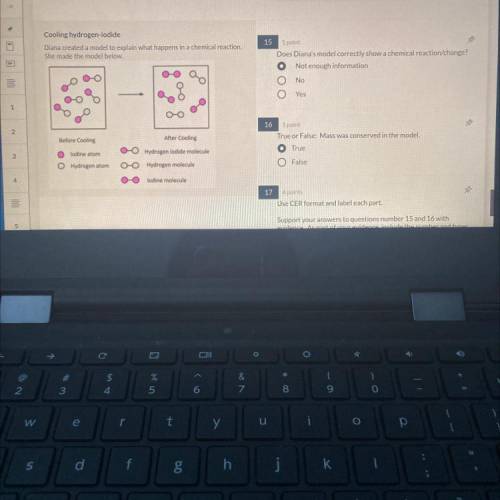

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, fantasticratz2
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 10:10, veronica022
Stage in which a typical star has completely stopped fusion
Answers: 1


Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Use CER format and label each part. Support your answers to question 15 and 16 with evidence. As par...
Questions in other subjects:



Social Studies, 12.04.2021 20:30



Mathematics, 12.04.2021 20:30



Mathematics, 12.04.2021 20:30




