
Chemistry, 26.02.2021 06:20 princessss30188
A 100 mL sample of ethanol at 25°C is mixed with a 300 mL sample of ethanol at -5°C. The mixture is allowed to come to thermal equilibrium. What is the final temperature?

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, ronny80
Supongamos que estás estudiando dos estrellas. ambas estrellas tienen la misma magnitud aparente, pero la estrella a tiene una magnitud absoluta mayor que la estrella b. ¿que puedes decir acerca de la distancia a la tierra de estas dos estrellas?
Answers: 3

Chemistry, 21.06.2019 21:00, alexmarche4675
Mrs. smith ordered a root beer float (vanilla ice cream + root beer). mrs. smith noticed that the three states of matter (solid, liquid, and gas) all existed simultaneously in her root beer float. a. identify each phase of matter in the root beer float. b. describe the particles of all three phases of matter in the root beer float. (how are the particles arranged and moving? ) c. identify one phase change you would see in a root beer float and described what causes this change.
Answers: 2

Chemistry, 22.06.2019 03:50, AysiaRamosLee
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 07:30, kimberlyrios12p0ts98
Which of the following best supports the concept that genetic information is passed on to offspring from both of their parents, not just one?
Answers: 2
You know the right answer?
A 100 mL sample of ethanol at 25°C is mixed with a 300 mL sample of ethanol at -5°C. The mixture is...
Questions in other subjects:


English, 02.08.2019 03:30


Geography, 02.08.2019 03:30


Mathematics, 02.08.2019 03:30



Physics, 02.08.2019 03:30

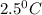
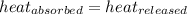
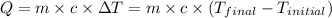
![m_1\times c_1\times (T_{final}-T_1)=-[m_2\times c_2\times (T_{final}-T_2)]](/tpl/images/1150/2880/09236.png) .................(1)
.................(1) = mass of first sample of ethanol = 100 ml
= mass of first sample of ethanol = 100 ml = mass of second sample of ethanol = 300 ml
= mass of second sample of ethanol = 300 ml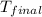 = final temperature = ?
= final temperature = ?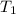 = temperature of first sample of ethanol =
= temperature of first sample of ethanol = 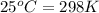
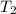 = temperature of second sample of ethanol =
= temperature of second sample of ethanol = 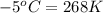
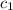 =
= 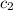 = specific heat of ethanol
= specific heat of ethanol![-100\times (T_{final}-298)=[300\times (T_{final}-268)]](/tpl/images/1150/2880/2eae3.png)




