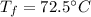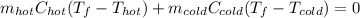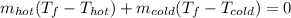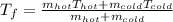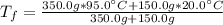Chemistry, 25.02.2021 08:10 rustalex6045
1. Mixing Water at Two Temperatures
a. One flask contains 150.0 g water at 20.0 °C. A second flask contains 350.0 g water at 95.0 °C. If the two water samples are mixed, what will the final temperature of the water be?
-Assume that the density of water it 1.000 g/cm^3
-The specific heat capacity for water is 4.184 J/g °C

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, chameleonsarelife
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3


Chemistry, 22.06.2019 18:00, jeepjose58
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1

Chemistry, 22.06.2019 19:30, gracieisweird12
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3
You know the right answer?
1. Mixing Water at Two Temperatures
a. One flask contains 150.0 g water at 20.0 °C. A second flask...
Questions in other subjects:


Mathematics, 20.07.2021 18:30


Mathematics, 20.07.2021 18:30




Mathematics, 20.07.2021 18:30