
Chemistry, 23.02.2021 21:10 deanlmartin
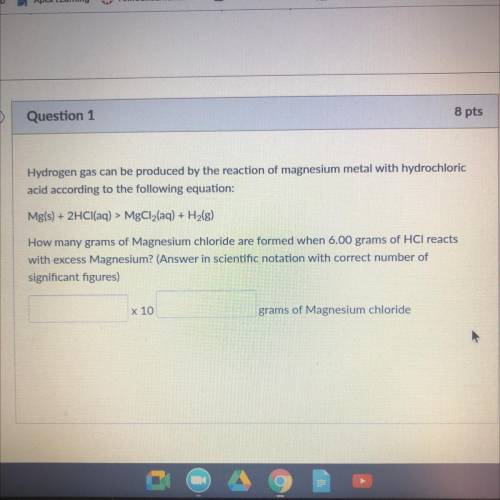
Hydrogen gas can be produced by the reaction of magnesium metal WICH
acid according to the following equation:
Mg(s) + 2HCl(aq) > MgCl2(aq) + H2(g)
How many grams of Magnesium chloride are Irmed when 6.00 grams of HCl reacts
with excess Magnesium? (Answer in scientific notation with correct number of
significant figures)
x 10
grams of Magnesium chloride

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, kellypechacekoyc1b3
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3


Chemistry, 22.06.2019 10:00, alexabdercmur
Why is the structure of molecule important to its function?
Answers: 1

Chemistry, 22.06.2019 13:00, devontemiles8868
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1
You know the right answer?
Hydrogen gas can be produced by the reaction of magnesium metal WICH
acid according to the followin...
Questions in other subjects:


Biology, 03.09.2020 01:01

Mathematics, 03.09.2020 01:01









