
Chemistry, 23.02.2021 01:40 xboxdude06
5.478 grams of potassium acetate and 2.143 grams of iron(III) hydroxide are added to a beaker containing 100.0 mL of water and stirred vigorously. A solid settles to the bottom of the beaker. If the water is decanted and the solid is dried, what is the maximum mass of solid that should be recovered

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, sillslola816oxb5h7
An aqueous solution of hydroiodic acid is standardized by titration with a 0.186 m solution of calcium hydroxide. if 26.5 ml of base are required to neutralize 20.3 ml of the acid, what is the molarity of the hydroiodic acid solution? m hydroiodic acid
Answers: 1

Chemistry, 22.06.2019 07:30, gwenparks
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

You know the right answer?
5.478 grams of potassium acetate and 2.143 grams of iron(III) hydroxide are added to a beaker contai...
Questions in other subjects:

Mathematics, 01.09.2021 19:10

English, 01.09.2021 19:10

Mathematics, 01.09.2021 19:10


Mathematics, 01.09.2021 19:10

Physics, 01.09.2021 19:10

Mathematics, 01.09.2021 19:10

Business, 01.09.2021 19:10

Mathematics, 01.09.2021 19:10

Biology, 01.09.2021 19:10

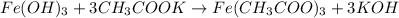
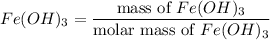
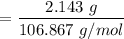
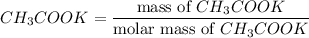
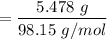
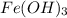 reacts with 3 mols of
reacts with 3 mols of 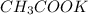
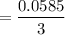 mol
mol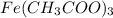 produced is 0.0195 mol
produced is 0.0195 mol

