
Chemistry, 22.02.2021 19:30 tejalawatson91
Which of the following are correct for zero-order reactions?
A. A higher concentration of reactants will not increase the reaction rate.
B. The units for the rate constant and the rate of reaction are the same.
C. The rate of reaction does not equal the rate constant.
D. The concentration of the reactants changes nonlinearly.
E. A zero-order reaction slows down as the reaction proceeds.

Answers: 1


Other questions on the subject: Chemistry


Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3

Chemistry, 22.06.2019 20:00, kalcius9698
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
Which of the following are correct for zero-order reactions?
A. A higher concentration of reactants...
Questions in other subjects:


English, 12.08.2020 07:01




Mathematics, 12.08.2020 07:01




Mathematics, 12.08.2020 07:01

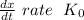 doesn't depend on reaction rate, a higher reaction rate does not intensify the reaction.
doesn't depend on reaction rate, a higher reaction rate does not intensify the reaction.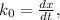 the created based and the reaction rate is about the same.
the created based and the reaction rate is about the same.

