
Chemistry, 18.02.2021 01:00 burritomadness
How do I make the molar masses for 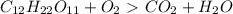 (reactant and products) equal? Please, tell me the steps and how to recalculate.
(reactant and products) equal? Please, tell me the steps and how to recalculate.

Answers: 3


Other questions on the subject: Chemistry


Chemistry, 21.06.2019 23:20, anggar20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

You know the right answer?
How do I make the molar masses for (reactant and products) equal? Please, tell me the steps and how...
Questions in other subjects:







Mathematics, 18.11.2020 23:10


Mathematics, 18.11.2020 23:10



