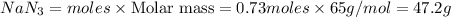Chemistry, 16.02.2021 04:20 baileyflemingde
Automotive airbags inflate when sodium azide decomposes explosively to its constituent elements. How many grams of sodium azide are required to produce 24.4 L of nitrogen gas at standard temperature and pressure? 2NaN3 --> 2Na + 3N2
47.2 g of sodium azide
106.2 g of sodium azide
1.63 g of sodium azide
0.726 g of sodium azide

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, ronny80
Supongamos que estás estudiando dos estrellas. ambas estrellas tienen la misma magnitud aparente, pero la estrella a tiene una magnitud absoluta mayor que la estrella b. ¿que puedes decir acerca de la distancia a la tierra de estas dos estrellas?
Answers: 3

Chemistry, 21.06.2019 23:00, fespinoza019
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1


You know the right answer?
Automotive airbags inflate when sodium azide decomposes explosively to its constituent elements. How...
Questions in other subjects:

Mathematics, 20.11.2019 23:31




Spanish, 20.11.2019 23:31



Mathematics, 20.11.2019 23:31


Mathematics, 20.11.2019 23:31

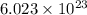 of particles.
of particles.
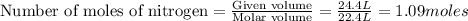
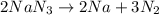
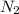 are produced by = 2 moles of
are produced by = 2 moles of 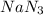
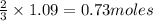 of
of