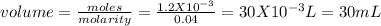Chemistry, 04.02.2020 12:58 abhibhambhani
20.0 ml of 0.06 m hcl (in a flask) is titrated with 0.04 m naoh (in a burette). how many milliliters of naoh needs to be used to reach the equivalence point?
a.0.03 ml
b.20.0 ml
c.20 ml
d.30 ml
2.which of the following would you identify a titration curve that involved a strong acid titrated by a weak base?
a. the ph at the equivalence point is lower than 7.
b. the ph at the equivalence point is higher than 7.
c. the titration curve begins at a higher ph and ends at a lower ph.
d. there is a rapid change in ph near the equivalence point (ph = 7).

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 12:00, KKHeffner02
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 23.06.2019 03:30, jennelledenise
Mr. rose asked his student to draw a quadrilateral with four unequal sides. an example of this kind of quadrilateral
Answers: 1
You know the right answer?
20.0 ml of 0.06 m hcl (in a flask) is titrated with 0.04 m naoh (in a burette). how many milliliters...
Questions in other subjects:

History, 04.08.2019 07:30

Chemistry, 04.08.2019 07:30


English, 04.08.2019 07:30


Social Studies, 04.08.2019 07:30



Physics, 04.08.2019 07:30

History, 04.08.2019 07:30