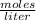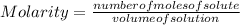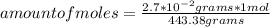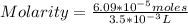2 points
2.7 x 10-2 grams of Pb(C2H302)4 are dissolved to make 3.5 mL of
solution. What is th...

Chemistry, 10.02.2021 14:00 andreamartinez7876
2 points
2.7 x 10-2 grams of Pb(C2H302)4 are dissolved to make 3.5 mL of
solution. What is the molarity?
Your Answer

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:40, draveon353
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 09:30, matpakootas521
Why do cells appear different in distilled water than they do in 10% salt water?
Answers: 2


Chemistry, 22.06.2019 16:50, TheOriginal2x
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3
You know the right answer?
Questions in other subjects:




Mathematics, 28.07.2019 05:00


Mathematics, 28.07.2019 05:00


Mathematics, 28.07.2019 05:00

English, 28.07.2019 05:00

English, 28.07.2019 05:00