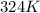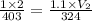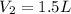Chemistry, 10.02.2021 09:10 maddie4985
If the initial temperature of a movable cylinder was 130 degrees Celsius
with a pressure of 1 ATM and an initial volume of 2 Liters, what would
the new volume be if the temperature was lowered to 324 degrees Kelvin
and the pressure was raised to 1.1 ATM?

Answers: 1


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, lakenyagillard79
Acurium-245 nucleus is hit with a neutron and changes as shown by the equation. complete the equation by filling in the missing parts. 52
Answers: 2

Chemistry, 22.06.2019 12:00, daytonalive83481
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 15:00, hockeykid7583
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 17:30, sheazy3709
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2
You know the right answer?
If the initial temperature of a movable cylinder was 130 degrees Celsius
with a pressure of 1 ATM a...
Questions in other subjects:


Mathematics, 04.06.2021 19:20


Mathematics, 04.06.2021 19:20


Chemistry, 04.06.2021 19:20


Chemistry, 04.06.2021 19:20


Mathematics, 04.06.2021 19:20


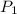 = initial pressure of gas = 1 atm
= initial pressure of gas = 1 atm
 = final pressure of gas = 1.1 atm
= final pressure of gas = 1.1 atm
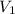 = initial volume of gas = 2 L
= initial volume of gas = 2 L
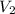 = final volume of gas = ?
= final volume of gas = ?
 = initial temperature of gas =
= initial temperature of gas = 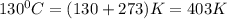
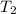 = final temperature of gas =
= final temperature of gas =