
Chemistry, 09.02.2021 01:00 eburnhisel2023
Given the following enthalpies of formation calculate the change in enthalpy for the reaction. Then justify if the reaction is endothermic or exothermic based on your calculations. PLEASE HELP WILL GIVE BRAINLIEST
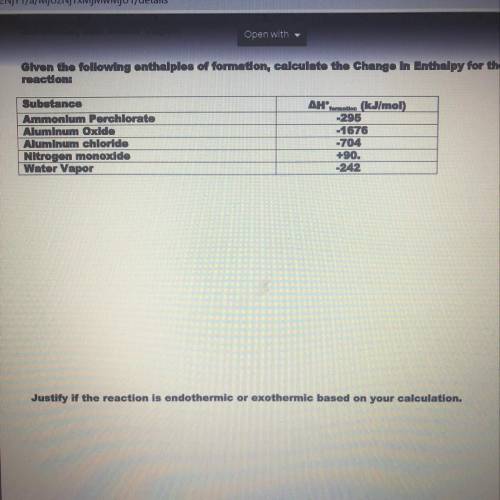

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, vlactawhalm29
Select the correct text in the passage. which sentences describe examples of sustainable living? i live in an old apartment building downtown, but my company is based in an office park on the outskirts of the city. i drive an old car that needs to be replaced. i plan to buy a hybrid for better gas mileage, but for now i am able to carpool with a couple of friends from work. the drive to the office park is about 45 minutes each way, but we do get to work in a modern building. the architects just received a leed certification for the design.
Answers: 3

Chemistry, 22.06.2019 06:30, reecedstceklein
Over the last 90 years, scientists have added to the body of evidence supporting the big bang theory. what is the latest piece of evidence discovered in 2014?
Answers: 1


You know the right answer?
Given the following enthalpies of formation calculate the change in enthalpy for the reaction. Then...
Questions in other subjects:



Mathematics, 17.02.2020 19:36







Mathematics, 17.02.2020 19:37



