Chemistry, 09.02.2021 01:00 thomasmurphy200
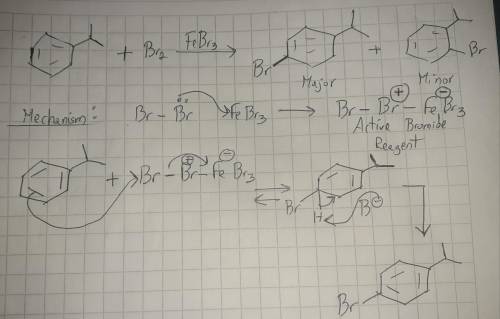
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropylbenzene. Complete the curved-arrow mechanism below, beginning with formation of the active brominating reagent. Remember to include lone pairs and formal charges where appropriate.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, only1cache
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3


Chemistry, 22.06.2019 05:40, yah2muchh
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 13:00, taylorpayne525p8qxky
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2
You know the right answer?
The electrophilic aromatic substitution of isopropylbenzene with FeBr3, Br2 gives 1-bromo-4-isopropy...
Questions in other subjects:

History, 15.10.2019 12:30

Mathematics, 15.10.2019 12:30

English, 15.10.2019 12:30


Mathematics, 15.10.2019 12:30

Mathematics, 15.10.2019 12:30

Mathematics, 15.10.2019 12:30


Mathematics, 15.10.2019 12:30