
Chemistry, 08.02.2021 22:00 carmenala2
A reaction that proceeds by first-order irreversible kinetics is oxidizing chemical A in a wastewater treatment basin with a mean residence time of 1.5 hours. The reaction rate constant is The basin is unbaffled and may be characterized as two completely mixed tanks in series. If the steady-state influent concentration is 30 mg/l, find the effluent concentration. If baffles are placed in the basin so that the basin may be characterized as four completely mixed tanks in series, and the mean residence time remains constant, find the effluent concentration.

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, cynthiagutierrez65
Where can i find naap lab answers sheet/key?
Answers: 1



Chemistry, 22.06.2019 18:30, rosie20052019
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1
You know the right answer?
A reaction that proceeds by first-order irreversible kinetics is oxidizing chemical A in a wastewate...
Questions in other subjects:





Mathematics, 21.05.2020 03:59


Mathematics, 21.05.2020 03:59



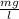
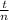 )ⁿ
)ⁿ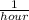 )
)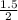 )²
)²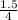 )⁴
)⁴

