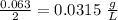Chemistry, 08.02.2021 19:00 amandasantiago2001
A chemistry student is given 2.00 L of a clear aqueous solution at 43.° C. He is told an unknown amount of a certain compound X is dissolved in the solution. The student allows the solution to cool to 25.° C. At that point, the student sees that a precipitate has formed. He pours off the remaining liquid solution, throws away the precipitate, and evaporates the water from the remaining liquid solution under vacuum. More precipitate forms. The student washes, dries and weighs the additional precipitate. It weighs 0.062 kg1) Using only the information above, can you calculate the solubility of X in water at 25 degrees C?2) If yes calculate it. Round answer to 2 significant digits

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, actheorian8142
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 05:00, YoEsMyles3115
0.2348 grams of pbcl2 used to form 44.0 ml of solution.
Answers: 1


Chemistry, 22.06.2019 10:00, nana54muller
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1
You know the right answer?
A chemistry student is given 2.00 L of a clear aqueous solution at 43.° C. He is told an unknown amo...
Questions in other subjects:




Social Studies, 13.08.2019 04:30






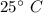 .
.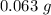 . They have a
. They have a 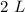 the solution, they may disregard the volume increases due to its precipitation. The intensity therefore is
the solution, they may disregard the volume increases due to its precipitation. The intensity therefore is