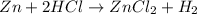Chemistry, 07.02.2021 21:30 HopeBordelon4
Calculate the mass of the zinc that reacts with 4.11 g of hydrochloric acid to form 9.1 g of zinc chloride and 3.97 g of hydrogen gas.

Answers: 2


Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, kayleg907436
Arollercoaster car at the top of a hill has potential energy kinetic energy chemical energy light energy
Answers: 1

Chemistry, 22.06.2019 02:00, lwattsstudent
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1


Chemistry, 22.06.2019 05:30, stellaglenn205
What reaction is taking place? 02 + c3h8 = h20 + co2
Answers: 1
You know the right answer?
Calculate the mass of the zinc that reacts with 4.11 g of hydrochloric acid to form 9.1 g of zinc ch...
Questions in other subjects:


Mathematics, 12.05.2021 14:00

English, 12.05.2021 14:00

Social Studies, 12.05.2021 14:00

Mathematics, 12.05.2021 14:00

Mathematics, 12.05.2021 14:00


English, 12.05.2021 14:00

SAT, 12.05.2021 14:00

Mathematics, 12.05.2021 14:00