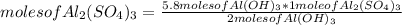Chemistry, 07.02.2021 20:10 salsaanddoritos899
Based on the equation below, how many moles of aluminum sulfate (Al2(SO4)3) will be produced from the reaction of 5.8 moles of aluminum hydroxide (Al(OH)3) and excess sulfuric acid (H2SO4)? 2Al(OH)3 + 3H2SO4 → Al2(SO4)3 + 6H2O

Answers: 3


Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:20, mgavyn1052
Calculate the molarity of the solution. 6.02 x 1022 molecules of hci (molecular weight = 36.5 g/mole) in 2.0 liters of water m
Answers: 1

Chemistry, 21.06.2019 23:00, daryondaniels28
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

You know the right answer?
Based on the equation below, how many moles of aluminum sulfate (Al2(SO4)3) will be produced from th...
Questions in other subjects:

Social Studies, 21.11.2020 02:40

Chemistry, 21.11.2020 02:40

History, 21.11.2020 02:40



Biology, 21.11.2020 02:40

Mathematics, 21.11.2020 02:40


Mathematics, 21.11.2020 02:40